More than 3,600 °C can sound unreal: standard values for this material sit around 3652–3697 °C (~6332 °F), a scale that shapes modern industry.
Here we define that range and explain why small differences appear across studies due to measurement setups and reference standards.
In oxygen-bearing air, oxidation begins near 700 °C, so high temperature operations usually run in inert or vacuum atmospheres to protect part integrity.
This carbon-based material endures heat, which makes it vital for refractories, electrodes, crucibles, rocket nozzles, and furnace parts used in steelmaking and aerospace.
Precision machining matters: tight tolerances and fine surface finish keep components reliable under extreme heat, which is a core capability of Rapidaccu for prototypes and production.
Later sections will unpack atomic structure, bonding energies, and how pressure, impurities, and atmospheres change behavior. For a quick scientific overview, see this summary on graphite on Wikipedia.
Graphite melting point at a glance: exact figures, ranges, and why they matter
When parts must survive extreme heat, exact thermal numbers drive design decisions.
Accepted values at standard atmospheric pressure
The accepted range sits at 3652–3697 °C (about 6332 °F). This very high band shows why this material suits furnaces, crucibles, and thermal protection parts.
Why reported figures vary
Different labs use varied apparatus, sample shapes, and purity levels. Test atmospheres—vacuum or inert gas versus air—also change results. Such factors explain small shifts inside the tight band engineers use.
Practical guidance for designers
- Match grade and geometry to service temperatures, heat flux, and dwell time.
- Account for oxidation above roughly 700 °C in air; use shielding or controlled gas.
- Plan thermal cycling to avoid surface recession and thermal shock damage.
| Condition | Accepted Range | Design Effect |
|---|---|---|
| Standard atmospheric pressure, inert gas | 3652–3697 °C (≈6332 °F) | Use full range for safety margins in thermal designs |
| Oxidizing air | Oxidation begins ≈700 °C | Require shielding; not suitable for open-air at high temperature |
| Variable purity or sample geometry | Apparent shifts within band | Confirm with supplier datasheets and lab tests |
| High thermal flux / cycling | Service-dependent | Specify machining tolerances to maintain function over cycles |
Rapidaccu supports teams that design around these values by delivering precision-machined parts that hold tolerances and surface finish through thermal cycles—from prototype to production.
From carbon atoms to covalent bonds: how graphite’s crystal structure drives its high melting point
At atomic scale, bond geometry and stacking define thermal resilience. Stacked graphene sheets form a layered crystal structure with sp2-hybridized carbon atoms. Each carbon atom links to three neighbors in hexagonal rings, creating strong in-plane covalent bonds and a sea of delocalized electrons that boost conductivity and stiffness.
An individual C–C bond within a layer measures about 0.142 nm, while layers sit roughly 0.335 nm apart. That short in-plane distance and long interlayer gap create marked anisotropy. In-plane covalent bonds demand large energy to break, which explains a very high melting point under inert conditions.
Strong covalent planes vs. weak interlayer forces
Van der Waals forces hold layers together. Those weak forces let sheets slide, giving lubricity and easier machining along planes despite strong in-plane bonds.
Alpha and beta stacking and thermal stability
Alpha (ABA) stacking is stable at service temperatures. Beta (ABC) can form under shear but tends to revert to alpha on heating near 1300 °C. This transition rarely affects high-temperature performance.

- In-plane covalent bonds set strength and thermal resistance.
- Interlayer forces enable cleavage and anisotropic machining.
- Design parts with layer orientation in mind to preserve edge integrity.
| Feature | Metric / Behavior | Design note |
|---|---|---|
| In-plane C–C bond | ≈0.142 nm — strong covalent | Align tooling to protect planes |
| Interlayer spacing | ≈0.335 nm — weak van der Waals | Expect easy shear and directional finish |
| Stacking | Alpha (ABA) stable; Beta (ABC) reverts on heat | Orient parts for thermal cycles |
Rapidaccu’s precision CNC workflows respect anisotropy in this crystal structure, choosing toolpaths and feeds that protect in-plane integrity and surface finish for reliable component performance.
whats is the graphite melting point under different conditions
High pressure, impurities, and atmosphere each change how graphite behaves near extreme temperatures.
Effect of pressure: atmospheric to ultra-high
At atmospheric pressure, reported values stay within a narrow band and design margins are predictable.
Under very high pressure, the layered structure compresses and interatomic interactions strengthen. That raises apparent melting values and can push phase behavior toward diamond or other regimes.
Impurity influence on lattice and bonds
Trace elements such as boron or nitrogen alter local bond energies and lattice coherence. Small additions can either raise or lower melting behavior depending on chemistry and concentration.
Specify grade purity to avoid unexpected shifts in performance at service temperatures.
Reactive atmospheres and oxidation above ~700 °C
Oxygen-containing gases drive surface loss above about 700 °C, so inert gas or vacuum is standard for high-temperature service.
Halogenated or oxidizing environments can cause rapid mass loss well before intrinsic melting, lowering practical survivability.
- Control atmosphere and gas purity during the test process.
- Choose high-purity grades to stabilize properties across conditions.
- Use finishes, radii, or coatings to reduce oxidation in service.
| Variable | Effect | Design guidance |
|---|---|---|
| Pressure | Raises melt behavior at high values | Specify expected pressure in material selection |
| Impurities (B, N) | Alters bond energies and lattice | Order certified purity and test samples |
| Atmosphere | Oxidation above ~700 °C causes mass loss | Use inert or vacuum and protective finishes |
For lab-to-field comparisons, report environment, dwell temperatures, and gas purity. Rapidaccu advises on design-for-environment, selecting grades and finishes that match pressure, atmosphere, and temperature conditions and machining parts to preserve properties. Learn more about graphite melt behavior in controlled tests graphite melt behavior.
Graphite vs. diamond: comparing melting points and crystal characteristics
Comparing carbon allotropes highlights how small changes in bond topology change thermal behavior.
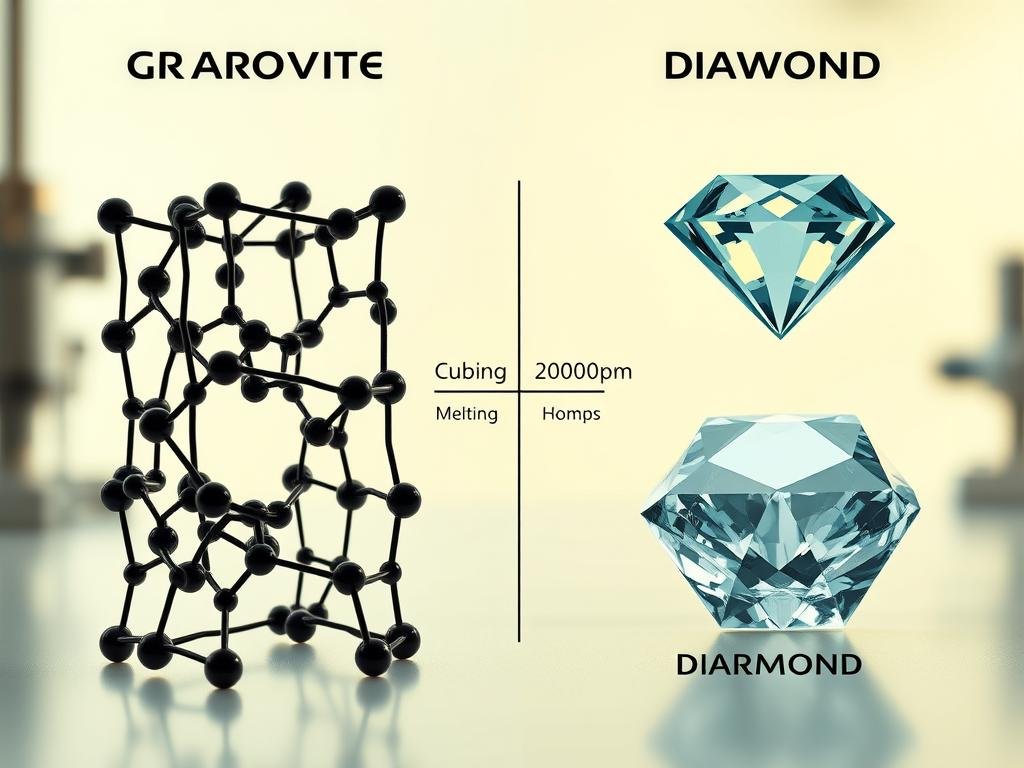
Diamond’s 3D covalent network
Diamond forms a tetrahedral sp3 lattice with four strong covalent bonds per atom. This fully connected crystal demands more energy to disrupt.
Layered sp2 structure
Layered sp2 networks have strong in-plane bonds but weak interlayer attraction. That contrast lowers required energy relative to a 3D network.
- Benchmark temperatures: graphite range ~3652–3697 °C versus diamond ≈4000 °C under suitable test conditions.
- More bonds per atom and no weak gaps explain diamond’s higher high melting point and stiffness.
- Graphite’s cleavage and machinability favor many thermal parts despite a slightly lower high melting advantage for diamond.
| Feature | Graphite (sp2) | Diamond (sp3) |
|---|---|---|
| Network | Layered sheets with weak interlayer forces | Three-dimensional tetrahedral lattice |
| Bonds per atom | Three strong in-plane covalent bonds | Four covalent bonds to neighbors |
| Typical benchmark temperature | ≈3652–3697 °C (atmospheric/inert) | ≈4000 °C (suitable conditions) |
| Practical note | Better machinability, cost-effective for components | Higher thermal limit, costly and hard to process |
Rapidaccu supports R&D teams with precision test coupons and fixtures to compare allotropes under controlled temperature and atmospheric pressure. This helps engineers quantify trade-offs and pick the right carbon structure for their application.
High-temperature applications that rely on graphite’s melting point
Industries rely on carbon parts that keep shape and function when exposed to sustained extreme temperatures. This section highlights key uses and how material choice affects process outcomes.
Graphite crucibles in metallurgy: purity, non-reactivity, and heat resistance
Crucibles hold molten metal without contaminating copper, iron, or alloys. Purity and chemical inertness preserve melt quality and support consistent process control.
Graphite blocks in metallurgical and resistance furnaces
Blocks line furnace interiors to manage heat flux and withstand thermal cycling. Their structure maintains dimensional stability under prolonged service.
Rocket nozzles and aerospace components
Rocket nozzles face extremely high gas velocities and temperatures. High melting capability and thermal conduction protect against localized failure in flight conditions.
Electrodes and refractories for steelmaking
Electrodes carry current and stand up to slag and heat in foundries. Refractories provide insulation and mechanical support at sustained high temperatures.
Battery anodes: performance for lithium-ion cells
As an anode material, graphite brings conductivity and stable intercalation. Precision shapes improve energy density and manufacturing yield.
Why precision machining matters: Rapidaccu’s CNC expertise
Tight tolerances, smooth edges, and controlled surface finish cut oxidation sites and stress risers. Rapidaccu’s 15+ years of CNC work on graphite, plastics, and metal optimizes toolpaths, dust handling, and QA for repeatable parts from prototype to production.
| Application | Key benefit | Specification guidance |
|---|---|---|
| Crucibles | Low contamination | High-purity grade, inert atmosphere |
| Furnace blocks | Thermal stability | Match grade to heat flux and cycling |
| Aerospace parts | Heat and erosion resistance | Design for airflow and protective coatings |
| Battery anodes | Conductivity and intercalation | Precision geometry, certified material |
- Specify expected pressure, atmospheres, and dwell temperatures.
- Choose grades and finishes that suit service conditions.
- Use precision machining to extend service life in high temperature applications.
Conclusion
A clear benchmark near 3652–3697 °C anchors material choices for high-temperature work. This range helps teams size safety margins and pick suitable grades.
Pressure, atmosphere, and trace impurities shift reported values and service stability. Oxidation above roughly 700 °C often limits practical life more than nominal numbers.
That behavior traces to carbon atoms arranged in layered networks with strong in-plane bonds. This structure gives high melting resilience while requiring careful environment control.
Specify atmospheres, temperatures, and pressure constraints in designs and test plans. For hands-on help, collaborate with Rapidaccu to prototype, validate, and scale parts that retain properties graphite offers, using precision CNC for repeatable geometry and finish. Learn more on melting point of graphite.
FAQ
What temperature range is commonly reported for graphite at standard atmospheric pressure?
Accepted experimental values cluster around 3652–3697 °C (about 6332 °F). Those figures come from high-temperature studies that heat pure carbon in inert conditions and measure when the solid lattice breaks down.
Why do reported values for this property sometimes differ between sources?
Results vary because tests use different methods, sample purities, and atmospheres. Measurements in vacuum, inert gas, or under pressure all shift outcomes. Equipment calibration and detection techniques also influence reported numbers.
How does carbon bonding within the crystal explain such high thermal resistance?
Carbon atoms form strong sp2 covalent bonds inside hexagonal layers. Those in-plane bonds require large energy to break, so the material endures extreme heat. Weak van der Waals forces hold layers together, so delamination can occur before full lattice breakdown.
What role does stacking order play in thermal stability?
Different stacking sequences, commonly labeled alpha and beta, change interlayer alignment and phonon behavior. These subtle structural differences slightly affect heat conduction and stability, but in-plane covalent bonding dominates melting behavior.
How does external pressure alter the temperature at which graphite transforms or melts?
Raising pressure can increase the temperature required for phase changes. Under extremely high pressure, carbon may convert toward diamond-like structures before melting, shifting the effective transition point compared with atmospheric conditions.
Can impurities change the thermal behavior of carbon materials?
Yes. Dopants such as boron or nitrogen distort the lattice and alter bond energies. Even small impurity levels can lower stability or promote graphitization at lower temperatures, changing practical performance in furnaces or crucibles.
At what temperatures does oxidation become a concern for carbon materials in air?
Oxidation accelerates above roughly 700 °C in oxygen-containing environments. Protective atmospheres or coatings are required for high-temperature service; otherwise, weight loss and structural degradation occur well below lattice breakdown.
How does diamond compare in thermal resistance and bonding?
Diamond features an sp3 tetrahedral network of covalent bonds, producing a different crystal and higher cohesive energy. Its thermal stability is often cited near 4000 °C under specific conditions, but phase behavior differs because of bonding geometry and conversion pathways under pressure.
Why do bonding types (sp2 vs. sp3) require different energies to disrupt?
Bond geometry affects bond strength and the number of bonds per atom. sp3 bonding links each carbon to four neighbors in a 3D network, while sp2 links three neighbors in planar sheets. Breaking or reorganizing those networks demands different energy inputs, so their high-temperature responses diverge.
What industrial uses depend on high-temperature stability of carbon materials?
Industries use high-purity carbon for crucibles in metallurgy, furnace linings, rocket nozzle components, and electrodes in steelmaking. The material’s thermal conductivity, chemical resistance in inert atmospheres, and machinability make it valuable.
How does machining quality affect performance in extreme-heat applications?
Precision machining ensures tight tolerances and surface integrity, reducing stress concentrators and oxidation sites. Expert CNC work improves lifetime and reliability for components in furnaces, aerospace parts, and electrochemical cells.
Are carbon materials used in batteries affected by these high-temperature properties?
Battery anodes leverage layered carbon structures for ion intercalation, not high-temperature tolerance. Still, the material’s electrical conductivity, structural stability, and purity influence cycle life and performance in lithium-ion cells.

